UNIT-I CHAPTER 2 HUMAN REPRODUCTION Chapter Outline 2.1 Human reproductive system 2.2 Gametogenesis 2.3 Menstrual cycle 2.4 Fertilization and implantation 2.5 Maintenance of pregnancy and embryonic development 2.6 Parturition and lactation Learning Objectives ► Creates an awareness …
UNIT-I CHAPTER 1 REPRODUCTION IN ORGANISMS
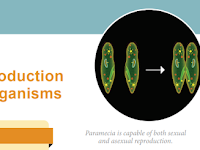
UNIT-I CHAPTER 1 Reproduction in Organisms Chapter Outline 1.1. Modes of reproduction 1.2. Asexual reproduction 1.3. Sexual reproduction Learning Objectives ➤ Learns the modes of asexual reproduction in organisms. ➤ Understands the significance of asexual reproduction. ➤ Comprehen…
Popular Posts
-
UNIT-I CHAPTER 2 HUMAN REPRODUCTION Chapter Outline 2.1 Human reproductive system 2.2 Gametogenesis 2.3 Menstrual cycle 2.4 Fertilizat...
-
UNIT-I CHAPTER 1 Reproduction in Organisms Chapter Outline 1.1. Modes of reproduction 1.2. Asexual reproduction 1.3. Sexual reproductio...
Powered by Blogger.
Labels
- 12 BIO ZOOLOGY (2)
Blog Archive
Recent Posts
Definition List
- REPRODUCTION IN ORGANISMS
- Living organisms show life involving birth, growth, development, maturation, reproduction and death. Reproduction is the fundamental feature of all living organisms. It is a biological process by which organisms produce their young ones. The young ones grow and mature to repeat the process. Thus reproduction results in continuation of species and introduces variations in organisms, which are essential for adaptation and evolution of their own kind.
- HUMAN REPRODUCTION
- Every organ system in the human body works continuously to maintain homeostasis for the survival of the individual. The human reproductive system is essential for the survival of the species. An individual may live a long healthy life without producing an offspring, but reproduction is inevitable for the existence of a species.
Featured Post
UNIT-I CHAPTER 2 HUMAN REPRODUCTION
UNIT-I CHAPTER 2 HUMAN REPRODUCTION Chapter Outline 2.1 Human reproductive system 2.2 Gametogenesis 2.3 Menstrual cycle 2.4 Fertilizat...